New Publication in Cell Chemical Biology
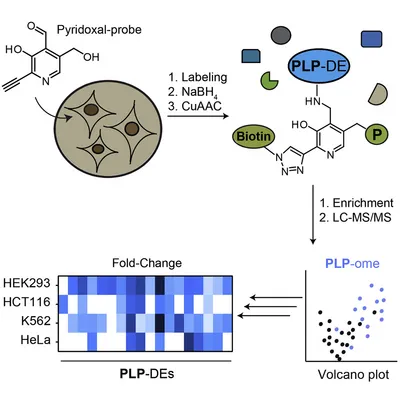
Pyridoxal 5′-phosphate (PLP) is a versatile cofactor that catalyzes a plethora of chemical transformations within a cell. Although many human PLP-dependent enzymes (PLP-DEs) with crucial physiological and pathological roles are known, a global method enabling their cellular profiling is lacking. Here, we demonstrate the utility of a cofactor probe for the identification of human PLP-binding proteins in living cells. Striking selectivity of human pyridoxal kinase led to a customized labeling strategy covering a large fraction of known PLP-binding proteins across various cancer-derived cell lines. Labeling intensities of some PLP-DEs varied depending on the cell type while the overall protein expression levels of these proteins remained constant. In addition, we applied the methodology for in situ screening of PLP-antagonists and unraveled known binders as well as unknown off-targets. Taken together, our proteome-wide method to study PLP-DEs in human cancer-derived cells enables global understanding of the interactome of this important cofactor.
Fux, A., Pfanzelt, M., Kirsch, V.C., Hoegl, A., Sieber, S.A., "Customizing functionalized cofactor mimics to study the human pyridoxal 5’-phosphate-binding proteome", Cell Chem. Biol. 2019, accepted
Link: 10.1016/j.chembiol.2019.08.003
Reproduced with permission.