New Publication in Chemical Communications
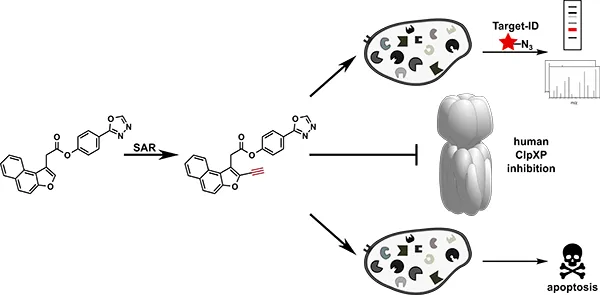
Human caseinolytic protease P (hClpP) is important for degradation of misfolded proteins in the mitochondrial unfolded protein response. We here introduce tailored hClpP inhibitors that utilize a steric discrimination in their core naphthofuran scaffold to selectively address the human enzyme. This novel inhibitor generation exhibited superior activity compared to previously introduced beta-lactones, optimized for bacterial ClpP. Further insights into the bioactivity and binding to cellular targets were obtained via chemical proteomics as well as proliferation- and mi gration studies in cancer cells
Gronauer, T.F., Mandl, M.M., Lakemeyer, M., Hackl, M.W., Meßner, M., Korotkov, V.S., Pachmayr, J., Sieber, S.A., "Design and synthesis of tailored human caseinolytic protease P inhibitors", Chem. Commun. 2018, accepted.
Link: 10.1039/C8CC05265D
Reproduced with permission from The Royal Society of Chemistry.