New Publication in ACS Infectious Diseases
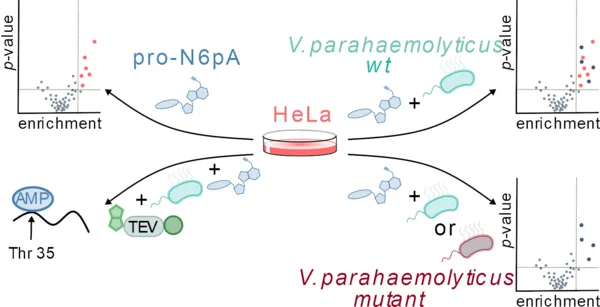
Bacteria utilize versatile strategies to propagate infections within human cells, e.g., by the injection of effector proteins, which alter crucial signaling pathways. One class of such virulence-associated proteins is involved in the AMPylation of eukaryotic Rho GTPases with devastating effects on viability. In order to get an inventory of AMPylated proteins, several technologies have been developed. However, as they were designed for the analysis of cell lysates, knowledge about AMPylation targets in living cells is largely lacking. Here, we implement a chemical-proteomic method for deciphering AMPylated host proteins in situ during bacterial infection. HeLa cells treated with a previously established cell permeable pronucleotide probe (pro-N6pA) were infected with Vibrio parahaemolyticus, and modified host proteins were identified upon probe enrichment and LC-MS/MS analysis. Three already known targets of the AMPylator VopS—Rac1, RhoA, and Cdc42—could be confirmed, and several other Rho GTPases were additionally identified. These hits were validated in comparative studies with V. parahaemolyticus wild type and a mutant producing an inactive VopS (H348A). The method further allowed to decipher the sites of modification and facilitated a time-dependent analysis of AMPylation during infection. Overall, the methodology provides a reliable detection of host AMPylation in situ and thus a versatile tool in monitoring infection processes.
Rauh, T., Brameyer, S., Kielkowski, P., Jung, K., Sieber, S. A., "MS-based in situ proteomics reveals AMPylation of host proteins during bacterial infection", ACS Infect. Dis.