Phosphinic Acid Functionalized Polyazacycloalkane Chelators for Radiodiagnostics and Radiotherapeutics: Unique Characteristics and Applications
Notni J, Šimeček J, Wester HJ
03.04.2014 [Original Artikel]
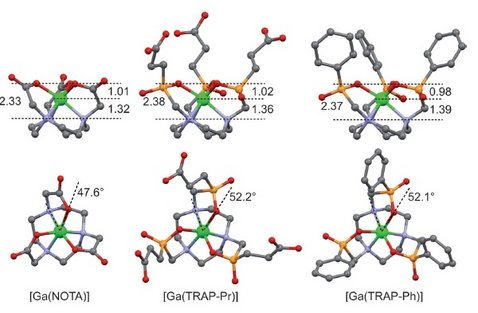
Given the wide application of positron emission tomography (PET), positron-emitting metal radionuclides have received much attention recently. Of these, gallium-68 has become particularly popular, as it is the only PET nuclide commercially available from radionuclide generators, therefore allowing local production of PET radiotracers independent of an on-site cyclotron. Hence, interest in optimized bifunctional chelators for the elaboration of 68Ga-labeled bioconjugates has been rekindled as well, resulting in the development of improved triazacyclononane-triphosphinate (TRAP) ligand structures. The most remarkable features of these ligands are unparalleled selectivity for GaIII, rapid GaIII complexation kinetics, extraordinarily high thermodynamic stability, and kinetic inertness of the respective GaIII chelates. As a result, TRAP chelators exhibit very favorable 68Ga-labeling properties. Based on the scaffolds NOPO (1,4,7-triazacyclononane-1,4-bis[methylene(hydroxymethyl)phosphinic acid]-7-[methylene(2-carboxyethyl)phosphinic acid]) and TRAP-Pr, tailored for convenient preparation of 68Ga-labeled monomeric and multimeric bioconjugates, a variety of novel 68Ga radiopharmaceuticals have been synthesized. These include bisphosphonates, somatostatin receptor ligands, prostate-specific membrane antigen (PSMA)-targeting peptides, and cyclic RGD pentapeptides, for in vivo PET imaging of bone, neuroendocrine tumors, prostate cancer, and integrin expression, respectively. Furthermore, TRAP-based 68Ga-labeled gadolinium(III) complexes have been proposed as bimodal probes for PET/MRI, and a cyclen-based analogue of TRAP-Pr has been suggested for the elaboration of targeted radiotherapeutics comprising radiolanthanide ions. Thus, polyazacycloalkane-based polyphosphinic acid chelators are a powerful toolbox for pharmaceutical research, particularly for the development of 68Ga radiopharmaceuticals.
Benefits of NOPO As Chelator in Gallium-68 Peptides, Exemplified by Preclinical Characterization of 68Ga-NOPO–c(RGDfK)
Šimeček J, Notni J, Kapp TG, Kessler H, Wester HJ
26.03.2014 [Original Artikel]
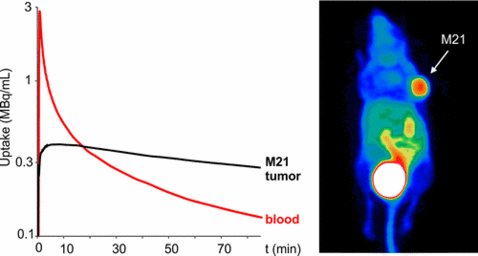
The αvβ3-integrin addressing cyclic pentapeptide cyclo(RGDfK) was conjugated to NOPO, 1,4,7-triazacyclononane-1,4-bis[methylene(hydroxymethyl)phosphinic acid]-7-[methylene(2-carboxyethyl)phosphinic acid], a bifunctional chelator with exceptional gallium-68 labeling properties. NOPO–c(RGDfK) and its Ga(III) and Cu(II) complexes showed high affinity to αvβ3integrin (IC50 = 0.94 ± 0.06, 1.02 ± 0.09, and 0.51 ± 0.06 nM, respectively). 68Ga labeling of NOPO–c(RGDfK) in an automated GMP-compliant procedure was performed with near-quantitative radiochemical yield, using precursor amounts as low as 0.5 nmol (approximately 0.6 μg). 68Ga-NOPO–c(RGDfK) was obtained with high purity (>99% by radio-HPLC/TLC) and, optionally, could be produced with specific activities up to 6 TBq/μmol. M21/M21L (human melanoma with high/low αvβ3 integrin expression) xenografted athymic CD-1 nude mice were used for biodistribution, in vivo stability studies, and PET imaging. 68Ga-NOPO–c(RGDfK) showed rapid and specific uptake in M21 tumor xenografts (2.02 ± 0.34% ID/g at 60 min p.i.) and was found stable in vivo. Its high hydrophilicity is reflected by an octanol–water distribution coefficient (log D = −4.6) which is more than 1 order of magnitude lower compared to respective NOTA or DOTA analogues. As expected, 68Ga-NOPO–c(RGDfK) thus showed fast renal clearance from nontargeted tissues. We conclude that NOPO might generally prove a useful means to improve renal clearance of corresponding radiopharmaceuticals by increasing the polarity of its bioconjugates. Favorable labeling properties render NOPO conjugates highly recommendable for reliable routine production of 68Ga-radiopharmaceuticals in a clinical setting.