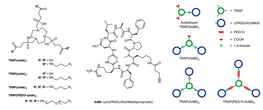
In-vivo PET imaging of the "cancer integrin" αvβ6 using gallium-68 labelled cyclic RGD nonapeptides
Notni J, Reich D, Maltsev OV, Kapp TG, Steiger K, Hoffmann F, Esposito I, Weichert W, Kessler H, Wester HJ
15.12.2016 [Original Artikel]
Expression of the cellular transmembrane receptor αvβ6 integrin is essentially restricted to malignant epithelial cells in carcinomas of a broad variety of lineages, while it is virtually absent in normal adult tissues. Thus, it is a highly attractive target for tumor imaging and therapy. Furthermore, αvβ6 integrin plays an important role for the epithelial-mesenchymal interaction and the development of fibrosis. On the basis of the Gallium-68 chelators TRAP (triazacyclononane-triphosphinate) and NODAGA, we synthesized mono- di- and trimeric conjugates of the αvβ6 integrinselective peptide cyclo(FRGDLAFp(NMe)K) via click chemistry (CuAAC). These were labeled with gallium-68 and screened regarding their suitability for in-vivo imaging of αvβ6 integrin expression by positron emission tomography (PET) and ex-vivo biodistribution in SCID mice bearing H2009 tumor (human lung adenocarcinoma) xenografts. For these, αvβ6 integrin expression in tumor and other tissues was determined by β6 immunohistochemistry (IHC). Despite the multimers showed higher αvβ6 integrin affinities (23–120 pM) than the monomers (260 pM), best results, that is, low background uptake and excellent tumor delineation, were obtained with the TRAP-based monomer 68Ga-Avebehexin. This compound showed the most favourable pharmacokinetics due to its high polarity (log D = –3.7) and presence of additional negative charges (carboxylates) on the chelator, promoting renal clearance. Although tumor uptake was low (0.65 ± 0.04 % injected dose per gram tissue, %ID/g), it was still higher than in all other organs except the kidneys, ranging from a maximum for the stomach (0.52 ± 0.04 % ID/g) to almost negligible for the pancreas (0.07 ± 0.01% ID/g). A low but significant target expression in tumor, lung and stomach was confirmed by IHC. Because of highly sensitive PET imaging even of tissues with low αvβ6 integrin expression density, we anticipate clinical applicability of 68Ga-Avebehexin for imaging of αvβ6 tumors and fibrosis by PET.
Variation of Specific Activities of 68Ga-Aquibeprin and 68Ga-Avebetrin Enables Selective PET Imaging of Different Expression Levels of Integrins a5b1 and avb3

Johannes Notni, Katja Steiger, Frauke Hoffmann, Dominik Reich, Markus Schwaiger, Horst Kessler, and Hans-Jürgen Wester
5.5.2016 [Original Artikel]
68Ga-aquibeprin and 68Ga-avebetrin are tracers for selective in vivo mapping of integrins α5β1 and αvβ3, respectively, by PET. Because both tracers exhibit high affinity to their respective targets, the aim of this study was to investigate the influence of the specific activity of preparations of both tracers on in vivo imaging results. Fully automated 68Ga labeling of 0.3 nmol of aquibeprin or avebetrin was done using buffered eluate fractions (600–800 MBq, pH 2) of an SnO2-based generator, affording the radiopharmaceuticals with specific activities greater than 1,000 MBq/nmol. Lower values ranging from 150 to 0.4 MBq/nmol were adjusted by addition of inactive compound (∼0.15–50 nmol) to the injected activity (∼20 MBq for PET, 5–7 MBq for biodistribution). For in vivo experiments, 6- to 12-wk-old female severe combined immunodeficiency mice bearing M21 xenografts (human melanoma, expressing both integrins α5β1 and αvβ3) were used. The expression density of integrin β3 was determined by immunohistochemistry on paraffin slices. For mass doses (specific activities) of less than 20 pmol (.1,000 MBq/nmol) and 1 nmol (20 MBq/nmol) per mouse, respectively, uptake of 68Ga-aquibeprin and 68Ga-avebetrin in M21 tumors dropped from 5.3 and 3.5 to 3.0 and 2.4 percentage injected dose per gram (%ID/g), respectively. When less than 20 pmol was applied, high uptake of 68Gaaquibeprin in the eyes (4.5 %ID/g) or 68Ga-avebetrin in adrenals (25.9 %ID/g), respectively, were found, which was reduced by 90% and 65% (0.44 and 6.2 %ID/g, respectively), for doses of 1 nmol. The highest tumor-to-tissue ratios were observed both in ex vivo biodistribution and PET for comparably large doses, for example, 6 nmol (0.65 mg/kg) 68Ga-aquibeprin per mouse (3.5 MBq/nmol). Presumably because of their high affinities, 68Ga-aquibeprin and 68Ga-avebetrin allow for selective addressing of target sites with different integrin expression levels by virtue of adjusting specific activity, which can be exploited for visualization of low-level target expression or optimization of tumor-tobackground contrast.
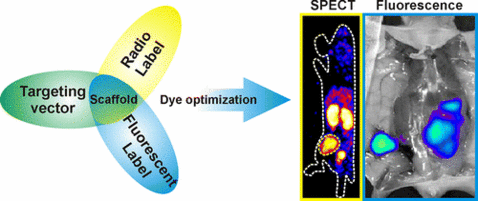
Tailoring Fluorescent Dyes To Optimize a Hybrid RGD-Tracer
Anton Bunschoten, Danny M. van Willigen, Tessa Buckle, Nynke S. van den Berg, Mick M. Welling, Silvia J. Spa, Hans-Jürgen Wester und Fijs W. B. van Leeuwen
13.04.2016 [Original Artikel]
Quantitative assessment of affinity and kinetics is acritical component in the development of (receptor-targeted)radiotracers. Forfluorescent tracers, such an assessment is currently not yet applied, while (small) changes in chemical composition of the fluorescent component might have substantial influence on the overall properties of a fluorescent tracer. Hybrid imaging labels that contain both a radiolabel and a fluorescent dye can be used to evaluate both the affinity (fluorescent label) and the in vivo distribution (radiolabel) of a targeted tracer. We present a hybrid label oriented and matrix-based scoring approach that enabled quantitative assessment of the influence of (overall) charge and lipophilicity of the fluorescent label on the (in vivo)characteristics of αvβ3-integrin targeted tracers. Systematic chemical alterations in the fluorescent dye were shown to result in aclear difference in the in vivo distribution of the different hybrid tracers. The applied evaluation technique resulted in an optimized targeted tracer for αvβ3-integrin, which combined the highest T/M ratio with the lowest uptake in other organs. Obviously this selection concept would also be applicable during the development of other (receptor-targeted) imaging tracers.
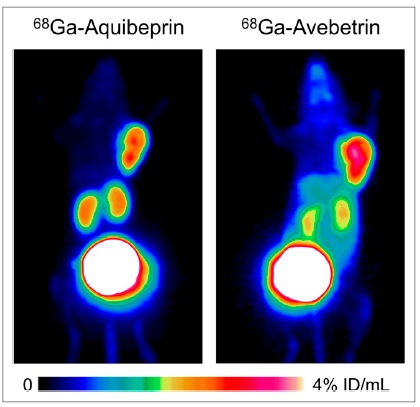
Complementary, Selective PET Imaging of Integrin Subtypes α5β1 and αvβ3 Using 68Ga-Aquibeprin and 68Ga-Avebetrin
Notni J, Steiger K, Hoffmann F, Reich D, Kapp TG, Rechenmacher F, Neubauer S, Kessler H, Wester HJ
01.03.2016 [Original Artikel]
Despite in vivo mapping of integrin αvβ3 expression being thoroughly investigated in recent years, its clinical value is still not well defined. For imaging of angiogenesis, the integrin subtype α5β1 appears to be a promising target, for which purpose we designed the PET radiopharmaceutical 68Ga-aquibeprin. 68Ga-aquibeprin was obtained by click-chemistry (CuAAC) trimerization of a α5β1 integrin–binding pseudopeptide on the triazacyclononane-triphosphinate (TRAP) chelator, followed by automated 68Ga labeling. Integrin α5β1 and αvβ3 affinities were determined in enzyme linked immune sorbent assay on immobilized integrins, using fibronectin and vitronectin, respectively, as competitors. M21 (human melanoma)-bearing severe combined immunodeficient mice were used for biodistribution, PET imaging, and determination of in vivo metabolization. The expression of α5 and β3 subunits was determined by immunohistochemistry on paraffin sections of M21 tumors. 68Ga-aquibeprin shows high selectivity for integrin α5β1 (50% inhibition concentration [IC50] = 0.088 nM) over αvβ3 (IC50 = 620 nM) and a pronounced hydrophilicity (log D = –4.2). Severe combined immunodeficient mice xenografted with M21 human melanoma were found suitable for in vivo evaluation, as M21 immunohistochemistry showed not only an endothelial and strong cytoplasmatic expression of the β3 integrin subunit but also an intense expression of the α5 integrin subunit particularly in the endothelial cells of intratumoral small vessels. Ex vivo biodistribution (90 min after injection) showed high uptake in M21 tumor (2.42 ± 0.21 percentage injected dose per gram), fast renal excretion, and low background; tumor-to-blood and tumor-to-muscle ratios were 10.6 ± 2.5 and 20.9 ± 2.4, respectively. 68Ga-aquibeprin is stable in vivo; no metabolites were detected in mouse urine, blood serum, kidney, and liver homogenates 30 min after injection. PET imaging was performed for 68Ga-aquibeprin and the previously described, structurally related c(RGDfK) trimer 68Ga-avebetrin, which shows an inverse selectivity for integrin αvβ3 (IC50 = 0.22 nM) over α5β1 (IC50 = 39 nM). In vivo target specificity was proven by cross-competition studies; tumor uptake of either tracer was not affected by the coadministration of 40 nmol (∼5 mg/kg) of the respective other compound. 68Ga-aquibeprin and 68Ga-avebetrin are recommendable for complementary mapping of integrins α5β1 and αvβ3 by PET, allowing for future studies on the role of these integrins in angiogenesis, tumor progression, metastasis, and myocardial infarct healing.